INNOVATIVE WATER SENSORS
While it sounds almost like science fiction, it’s a real water safety test, the brainchild of Chicago-based Stemloop’s founder and CEO, Khalid K. Alam, also an innovator for Chain Reaction Innovations (CRI) Cohort 3. CRI is a two-year entrepreneurship program at the U.S. Department of Energy’s Argonne National Laboratory, which embeds innovators in the Lab to help them grow their early-stage technologies.
“Our focus is on lead sensing, but we can detect a variety of heavy metals, pollutants, pesticides, pharmaceuticals, etc.,” Alam said. “If nature can sense it, we can turn it into one of our sensors. We are also developing a pathogen detection platform with a focus on COVID-19, but it can also apply to Legionella and other pathogens of concern.”
The same compounds that are toxic to humans are also toxic to microorganisms, Alam explained.
Alam, an RNA synthetic biologist, has his Ph.D.in Biochemistry, along with a Graduate Certificate in Life Science Innovation and Entrepreneurship, both from the University of Missouri.
ABOUT STEMLOOP
Stemloop was named after stem-loop, the core structure that RNA forms. RNA, DNA, and proteins play a role in Stemloop’s water test. There is also a double meaning to the company’s name, STEM (Science, Technology, Engineering, and Mathematics) and “loop,” which gives a nod to downtown Chicago’s Loop. The company has two faculty co-founders, Julius Lucks (whom Alam did a postdoctoral fellowship with at Northwestern University) and Michael Jewett (collaborator and director of the Center for Synthetic Biology at Northwestern).
Since joining Cohort 3 in June 2019 as one of its five innovators, Alam has achieved several milestones that include being awarded Small Business Innovation Research (SBIR) funding from the National Science Foundation in the amount of $256,000 and the National Institute of Environment Health Science in the amount of $225,000. This funding helped Alam reach another milestone, hiring full-time staff at Stemloop for the first time.
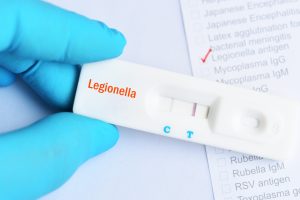
LEGIONELLA CHALLENGE
Stemloop’s first products will be focused on the detection of Legionella bacteria to address the long-standing challenge of healthcare-associated Legionella disease. Legionella thrive in building water systems that are not managed well due in part to lack of robust real-time testing capabilities.
“Our focus is on lead sensing, but we can detect a variety of heavy metals, pollutants, pesticides, pharmaceuticals, etc.,” Alam said.
While embedded at the Lab in building 446, Alam has worked with Argonne scientists Gyorgy Babnigg, a bioinformatician/molecular biologist at the Biosciences Division, and Andrzej Joachimiak, Director of Structural Biology Center and Midwest Center for Structural Genomics, “both of whom have deep expertise in the structure and function of ‘biosensor proteins’,” Alam said.
HOW IT WORKS
To create the water safety test, Stemloop first had to determine which microorganisms sense the toxins and pathogens that they are interested in detecting, which can vary due to the environment of the microorganism. E. coli, for example, does not typically sense lead because it usually lives in the gut and is not exposed to heavy metals. However, there are bacteria that have evolved in other environments that do sense lead, such as Staphylococcus aureus. Bioinformatics computer software is then used to understand biological data and to identify the sequence of DNA in the microorganism that encodes for the sensor.

Once this is determined, the DNA sequence from the microorganism (that encodes for the protein sensor sensing the toxin or pathogen) is synthesized. While synthetic DNA used to be very expensive a decade ago—potentially tens of thousands of dollars—today the cost is much more reasonable, Alam said.
“This is the stuff that life is made out of,” Alam pointed out.
Stemloop usually uses E. coli to produce the protein sensors because it is easy to work with, will grow fast, and there is a good understanding of how it functions. As noted earlier, E. coli, however, does not have certain protein sensors, such as for lead. So Stemloop adds the synthetic DNA, such as from Staphylococcus aureus, into E. coli and coaxes it to produce large amounts of the protein sensor. While Staphylococcus aureus senses lead, is not as easy to work with and has safety risks, so its synthetic DNA is used in lieu.
DNA codes for RNA, RNA codes for protein, and the protein then senses the toxin or pathogen, Alam said.
If the protein senses a pathogen or toxin, a reaction occurs, and the water being tested in the solution will change color.
IMMEDIATE RESULTS
“Our test is inexpensive and easy to use—just add a drop of water to test and wait for a color change,” Alam said. “The alternative is sending samples to a laboratory, spending upwards of a hundred bucks, and waiting days, if not weeks, for a single piece of data.”

